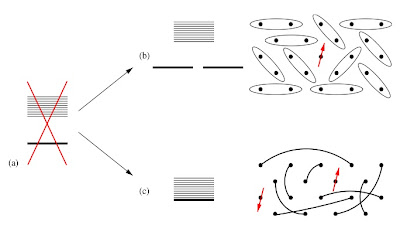
Previously I posted about recent numerical work which suggested that the Hubbard model on the honeycomb lattice and the Heisenberg model on the kagome lattice may have spin liquid ground states. At the Journal Club for Condensed Matter Physics, Steve Kivelson has a nice commentary on these two papers. One point important question he focuses on is whether or not these states have topological order, signified by a degenerate ground state, whose degeneracy only depends on the topology of the lattice [e.g. a torus].
Monday, January 31, 2011
Sunday, January 30, 2011
Do biologists and physicists need each other?
Do biologists really need physicists and mathematicians? Will the need increase in the next few decades? What are the landmark contributions of quantitative methods to biology? Seth Olsen brought to my attention an article, Mathematics is Biology's Next Microscope, Only Better; Biology is Mathematics Next Physics, Only Better by Joel E. Cohen.
It is worth reading, especially because it has lists of
- Mathematics arising from Biological Problems [Important historical contributions]
- 5 Biological challenges that could stimulate, and benefit from, major innovations in mathematics
- 5 Mathematical challenges that would contribute to the progress of Biology
- Important references.
Here are a few random comments.
I am not sure I agree with the title. I would say the point is more:
Quantitative methods are Biology's next microscope.
Biological physics is the next playground for emergent concepts and model.
Quantitative biology may lead to some interesting new applied mathematics.
It is a worry that the vast majority of the list of mathematical contributions to biological problems involves contributions from more than 50 years ago.
Saturday, January 29, 2011
Back up now!
The recent floods in Brisbane highlighted to me how we always need to be prepared for low probability-high impact events. So I went out and bought a second hard drive for backup. I realised that having a laptop and backup in the same location is not sufficient. Fires, floods, and theft do happen. So don't just back up now. Get a second one in a different location. Furthermore, the earlier you are in your career the more important this is. Losing your thesis or all your data is the worst thing that can happen.
Friday, January 28, 2011
Towards the spin Bose metal
As discussed in earlier posts and this review a strong candidate two-dimensional quantum spin model to have a spin liquid ground state is the Heisenberg model on the anisotropic triangular lattice with some ring exchange K. This model is directly relevant to the Mott insulating state of various charge transfer salts.
Studying such frustrated two-dimensional models numerically is a major challenge, and getting definitive results is currently not possible [see e.g. my post about recent work on the Kagome lattice model]. In contrast in one dimension the density matrix renormalisation group (DMRG) gives definitive results. Hence, one strategy is then to look at coupled chains in the hope that they capture essential physics.
There is a nice preprint by Block, Sheng, Motrunich, and Fisher which looks at the problem on a four chain system using the density matrix renormalisation group (DMRG). They find that once the ring exchange interaction K is larger than J/5 [quite a physically realistic value] that the ground state is a spin liquid: the spin Bose metal (SBM) proposed by Motrunich. They also studied the effect of spatial anisotropy and produced the phase diagram below.
It is fascinating that the organic charge transfer salts have parameter values that are approximately in the region where all three different phases can occur.
I thank H.-H Lai for bringing this preprint to my attention.
Studying such frustrated two-dimensional models numerically is a major challenge, and getting definitive results is currently not possible [see e.g. my post about recent work on the Kagome lattice model]. In contrast in one dimension the density matrix renormalisation group (DMRG) gives definitive results. Hence, one strategy is then to look at coupled chains in the hope that they capture essential physics.
There is a nice preprint by Block, Sheng, Motrunich, and Fisher which looks at the problem on a four chain system using the density matrix renormalisation group (DMRG). They find that once the ring exchange interaction K is larger than J/5 [quite a physically realistic value] that the ground state is a spin liquid: the spin Bose metal (SBM) proposed by Motrunich. They also studied the effect of spatial anisotropy and produced the phase diagram below.
It is fascinating that the organic charge transfer salts have parameter values that are approximately in the region where all three different phases can occur.
I thank H.-H Lai for bringing this preprint to my attention.
Thursday, January 27, 2011
A physicists view of hydrogen bonding
This afternoon I gave a talk at the cake meeting, "Empirical valence bond models for hydrogen bonding and proton transfer". The notes are here.
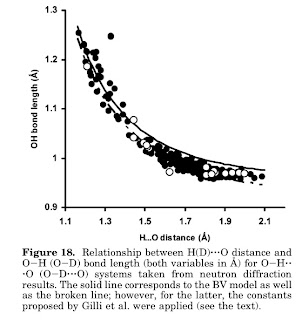
The figure below taken from a Chemical Reviews is interesting. It shows how for a
O...H-O system the correlation been O-H bond length and the O..H bond length has a natural explanation in terms of bond order conservation. To me this shows that hydrogen bonds are essentially a resonance between a covalent bond and a charge transfer excitation, as proposed by Coulson. i.e. the wavefunction is a superposition of A-H... B and A-...+H-B.
Wednesday, January 26, 2011
I need to write more negative posts!
My New Years wish for this blog is that it would attract more comments online. It now has several hundred pageviews per day, but rarely generates much discussion; except when it is occasionally provocative. It turns out there is some science to this. Read below a post Negatives attract from another blogger
while doing some research for a piece in an upcoming Nature Chemistry column on the roll of blogging in chemistry (due out in March), I ran across a fascinating paper in the European Physical Journal B: "Networks and emotion-driven user communities at popular blogs." The authors mathematically analyzed the topology (connectedness) of a large network of blogs and posts, assessing the emotional content along the way. (If the word bipartite doesn't scare you, read the paper.) They noted that large communities grow around posts with very negative language - and in the absence of such posts, there ends up being far less "cross-talk" between blogs. Nasty posts acted as glue.
Their data suggests that being negative also makes you popular in the blog world. More negatively worded posts attract more comments. Negative language in the comments attract more comments. Being obnoxious acts as a amplifier.
When I look at blogs — on any topic, science, theology, parenting — that provoke rather than explore or reflect, I've noticed that they tend to have many comments. (A couple come to mind, but I find myself reluctant to name names…though it strikes me it would be an interesting experiment to stir things up and see what happens to my comment box!)
Tuesday, January 25, 2011
Simplifying job applications
From the perspective of all concerned [job applicants, employers, reference letter writers] the mechanics of the delivery and collation of letters of reference for job applicants can be cumbersome. This process will be made somewhat easier by a new site AcademicJobsOnline.
The only glitch I found is that when using Safari on a Mac it would not allow me to upload a letter. I tried repeatedly. I then found it worked on a Windows machine. Hopefully this was just a problem with my machine and/or it will be fixed soon.
The only glitch I found is that when using Safari on a Mac it would not allow me to upload a letter. I tried repeatedly. I then found it worked on a Windows machine. Hopefully this was just a problem with my machine and/or it will be fixed soon.
Monday, January 24, 2011
A brief primer on diabatic states
Previously I discussed the importance of finding diabatic states as an alternative way to describe the electronic structure and potential energy surfaces of molecules. Here are a few key points.
In general if one knows the Born-Oppenheimer [i.e. adiabatic] eigenstates it is impossible to find perfectly diabatic states [i.e. ones which do not change with geometry] for a specific molecular system.
Hence, one must find approximately diabatic states. There are several alternative strategies.
a. Use chemically intuitive basis states such as valence bond structures [see for example].
b. Define states which constrain some physical observable, e.g., localisation of charge or bond length alternation.
c. The generalised Mulliken-Hush approach [due to Cave and Newton] is a specific case of b. One chooses the diabatic states so that the transition dipole moment between them vanishes. In different words, for a two-state system one evaluates the matrix elements of the dipole operator, with respect to the adiabatic eigenstates and then diagonalises this matrix. The basis states which diagonalise this matrix are then taken to be the diabatic states.
d. One block diagonalises the adiabatic Hamiltonian matrix according to some "minimisation" criteria. [This was pioneered by Cederbaum, Domcke, and collaborators].
See for example.
In general if one knows the Born-Oppenheimer [i.e. adiabatic] eigenstates it is impossible to find perfectly diabatic states [i.e. ones which do not change with geometry] for a specific molecular system.
Hence, one must find approximately diabatic states. There are several alternative strategies.
a. Use chemically intuitive basis states such as valence bond structures [see for example].
b. Define states which constrain some physical observable, e.g., localisation of charge or bond length alternation.
c. The generalised Mulliken-Hush approach [due to Cave and Newton] is a specific case of b. One chooses the diabatic states so that the transition dipole moment between them vanishes. In different words, for a two-state system one evaluates the matrix elements of the dipole operator, with respect to the adiabatic eigenstates and then diagonalises this matrix. The basis states which diagonalise this matrix are then taken to be the diabatic states.
d. One block diagonalises the adiabatic Hamiltonian matrix according to some "minimisation" criteria. [This was pioneered by Cederbaum, Domcke, and collaborators].
See for example.
Sunday, January 23, 2011
Seeing self-assembly
I discovered a real cool household experiment that can be used to demonstrate the complexity of interactions and structures that are present in a protein solution (milk).
Friday, January 21, 2011
Perfectly unproductive
Previously I posted about the issue of perfectionism and procrastination in academia. Hence, I was intrigued to see that the most read article on the University Affairs website is Perfectionist professors have lower research productivity.
Thursday, January 20, 2011
Should you do a postdoc?
Only if you have realistic expectations of what it may lead to. For the average postdoc there is only a small likelihood that it will lead to a permanent academic position. It is worth looking at the talk Challenges being faced by postdoctoral scholars in Canada. (Is it an "academic parking lot"?). Although focussed on Canada most of the issues are relevant world wide.
Ultimately, this is a very personal decision dependent on your own values and goals. But it should be a well informed decision.
Ultimately, this is a very personal decision dependent on your own values and goals. But it should be a well informed decision.
Wednesday, January 19, 2011
Jahn-Teller without symmetry
Generally, one only thinks of the Jahn-Teller effect in the context of systems which have certain discrete symmetries leading to degenerate energy eigenvalues. However, one can take a different perspective on the applicability of the theoretical machinery which has been developed for treating such problems. This point is made at the beginning of an extensive review by Bersuker:
This point is particularly important because of the ubiquity of conical intersections on potential energy surfaces for excited states of organic molecules.
I begin this review with a statement that the so-called Jahn-Teller (JT) effect (hereafter JTE), pseudo- JTE (PJTE), and Renner-Teller effect (RTE), jointly JT vibronic coupling effects, are not limited to a restricted variety of molecular polyatomic systems in specific electronic states, as it is often presented in the literature. In fact... the JT vibronic coupling theory is an approach to (a tool for) solving molecular and crystal problems, which is in principle applicable to any system with more than two atoms. Practically, this approach is most efficient when there are two or more coinciding (degenerate) or relatively close in energy (pseudo-degenerate) electronic states that become sufficiently strongly mixed when the nuclei displace from their initial reference configuration.
This point is particularly important because of the ubiquity of conical intersections on potential energy surfaces for excited states of organic molecules.
Tuesday, January 18, 2011
Incipient charge order destroys quasi-particles
A nice PRL just appeared by Lara Cano-Cortes, Jaime Merino, and Simone Fratini, Quantum critical behaviour of electrons at the edge of charge order. They consider an extended Hubbard model at one-quarter filling. As the intersite Coulomb repulsion V increases there is a quantum phase transition from a metallic Fermi liquid phase to a charge ordered phase. The striking finding is that the coherence temperature, below which quasi-particles exist, approaches zero as the transition is approached.
I also found the paper interesting because this model is directly relevant to a class of organic charge transfer salts, as discussed in a PRB by myself, Jaime, Brad Marston, and Oleg Sushkov.
I also found the paper interesting because this model is directly relevant to a class of organic charge transfer salts, as discussed in a PRB by myself, Jaime, Brad Marston, and Oleg Sushkov.
Monday, January 17, 2011
What has quantum chemistry really achieved?
A small part only of the body of contributions of quantum mechanics to chemistry has been purely quantum-mechanical in character; only in a few cases, for example, have results of direct chemical interest been obtained by the accurate solution of the Schrodinger wave equation... The principal contribution of quantum mechanics to chemistry has been the suggestion of new ideas, such as the resonance of molecules among several electronic structures with an accompanying increase in stability.Linus Pauling, Preface to The Nature of the Chemical Bond, First edition 1938
I wonder if this is still true today. Most computational chemists would bridle at that suggestion. But, I suspect that Pauling's point could still be argued today. Quantum mechanics has introduced important qualitative concepts such as potential energy surfaces, transition states, conical intersections, hybrid orbitals, ligand field theory, frontier orbitals, selection rules, .... These concepts are of far greater significance and success than the results of any detailed computations. Indeed the latter are largely of use to validate (and learn the boundaries of validity) of these concepts.
I welcome the views of readers.
Sunday, January 16, 2011
Are your results reproducible?
Reproducibility is meant to be basic tenet of science. Explicitly, a scientific paper is expected to report the results of experiments or calculations so that another scientist can reproduce them. This may sound very basic. But I fear this is beginning to be lost, particularly in papers that are computationally intensive and involve systems with many degrees of freedom.
A colleague was commenting to me recently that it has taken his group a very long time to reproduce the details of the calculations of another (prominent) group concerning a specific protein. The papers are sparse on details (such as details of basis sets, convergence criteria, starting geometries, ...). It turned out that the reason it was hard to reproduce the results was that the calculations actually were not very well converged.
This should not be. It is no excuse to claim that such details will make the paper too long or not be of interest to the average reader. Such technical details do not have to be in the actual paper. It is possible to deposit supplementary material on journal websites.
So when you are writing a paper make sure that all parameters you use in any calculation are specified. The same applies to papers mostly involving analytical calculations. All parameters and equations used to produce graphs need to be specified.
If you are refereeing a paper make sure it has enough detail. Otherwise it should not be published.
It is interesting that this point is made by Fritz Schaefer in his requirements for first drafts for papers from his group.
I realise I also need to be more diligent about this as well.
A colleague was commenting to me recently that it has taken his group a very long time to reproduce the details of the calculations of another (prominent) group concerning a specific protein. The papers are sparse on details (such as details of basis sets, convergence criteria, starting geometries, ...). It turned out that the reason it was hard to reproduce the results was that the calculations actually were not very well converged.
This should not be. It is no excuse to claim that such details will make the paper too long or not be of interest to the average reader. Such technical details do not have to be in the actual paper. It is possible to deposit supplementary material on journal websites.
So when you are writing a paper make sure that all parameters you use in any calculation are specified. The same applies to papers mostly involving analytical calculations. All parameters and equations used to produce graphs need to be specified.
If you are refereeing a paper make sure it has enough detail. Otherwise it should not be published.
It is interesting that this point is made by Fritz Schaefer in his requirements for first drafts for papers from his group.
I realise I also need to be more diligent about this as well.
Friday, January 14, 2011
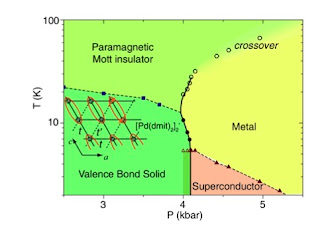
Review article on frustrated organics
Ben Powell and I have finished the revised and updated version of our review article, Quantum frustration in organic Mott insulators: from spin liquids to unconventional superconductors, which we have resubmitted to Reports in Progress in Physics. We welcome any comments and hope it will stimulate more work on this fascinating subject.
The flood at UQ
The flood waters are beginning to subside in Brisbane. Above is a picture of the northside of the UQ campus. Fortunately, no major buildings were affected; mostly sporting facilities and car parks were inundated.
Thanks again to colleagues from around the world who enquired about my well being. My house was fine. Mind you, the water got to within 100 metres. The route I normally walk with my son to school was underwater.
It is disturbing to read in the Australian, Alarming report on Brisbane River risks covered up, about how a 1999 report to the city council was kept secret until it was leaked years later. Why the secrecy? Too many developers and real estate agents wanted to make money by building and selling on low-lying land.....
Any lessons for scientists? Yes. I believe that when there are large amounts of money (funding) and power (prizes and careers) at stake it is hard for the potential beneficiaries to be objective about the truth. They will also be reluctant to want all the uncertainties about what is known or not known to come to light.
Wednesday, January 12, 2011
Excitation spectra in a spin liquid
Any rigorous theorem on quantum many-body systems is useful.
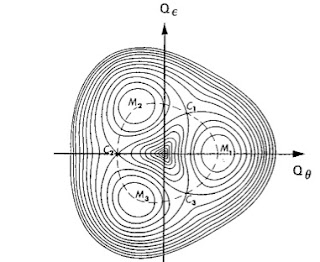
An important theorem for quantum spin systems was proven by Hastings. It is a generalization of the Lieb–Schultz–Mattis theorem to dimensions larger than one. It concerns spin-1/2 systems with one spin per unit cell on a two-dimensional lattice with periodic boundary conditions (on a torus). The theorem states that if there is no symmetry breaking then the ground state is separated from the first excited state by an energy gap that vanishes in the thermodynamic limit. Hence, under these specific conditions one cannot have a singlet ground state with a non-zero energy gap.
There is a nice discussion of the theorem in some lecture notes by Matthew Fisher, which contains the Figure below.
A hydrated university
This is a picture of the University of Queensland in 1974 when Brisbane had a major flood. A similar flood is happening right now!
I don't anticipate any problems with major buildings and labs because the university has exercised restraint and not built on areas that were flooded.
Thanks to my many overseas colleagues who have asked after my well-being. Fortunately, our house is high enough we will probably only have water in the back yard.
Saturday, January 8, 2011
A ladder towards two dimensions
Understanding quantum lattice models in two-dimensions is a major theoretical challenge.
Ladder models provide a means to investigate in a controlled manner (e.g., via DMRG, bosonisation, and weak-coupling renormalisation group) physics which it is hoped may be related to what occurs in the two-dimensional limit.
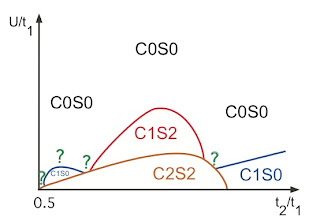
One can characterise different ground states on ladders by n and m, the number of gapless charge and spin modes, respectively. This leads to the notation CnSm and the following identifications.
C2S2 is the ladder analogue of Fermi liquid metal,
C1S0 is a superconductor,
C1S2 is a spin Bose metal,
C0S0 is a spin gapped Mott insulator,
C0S2 is a spin liquid Mott insulator.
Balents and Fisher used a weak-coupling RG approach to show that at half filling a two-leg ladder without frustration has a C0S0 ground state, which upon doping changes has a C0S0 ground state, which upon doping changes to C1S0 consistent with Anderson's RVB ideas.
Lai and Motrunich recently performed a weak-coupling renormalisation group analysis has been performed on the zig-zag ladder with longer-range Coulomb repulsion at half-filling. This is the ladder analogue of the Hubbard model that should describe organic charge transfer salts. The longer-range interaction stabilises the C2S2 phase and leads to a subtle competition between all the different phases listed above. Indeed it is interesting to compare their Figure 8
Ladder models provide a means to investigate in a controlled manner (e.g., via DMRG, bosonisation, and weak-coupling renormalisation group) physics which it is hoped may be related to what occurs in the two-dimensional limit.
One can characterise different ground states on ladders by n and m, the number of gapless charge and spin modes, respectively. This leads to the notation CnSm and the following identifications.
C2S2 is the ladder analogue of Fermi liquid metal,
C1S0 is a superconductor,
C1S2 is a spin Bose metal,
C0S0 is a spin gapped Mott insulator,
C0S2 is a spin liquid Mott insulator.
Balents and Fisher used a weak-coupling RG approach to show that at half filling a two-leg ladder without frustration has a C0S0 ground state, which upon doping changes has a C0S0 ground state, which upon doping changes to C1S0 consistent with Anderson's RVB ideas.
Lai and Motrunich recently performed a weak-coupling renormalisation group analysis has been performed on the zig-zag ladder with longer-range Coulomb repulsion at half-filling. This is the ladder analogue of the Hubbard model that should describe organic charge transfer salts. The longer-range interaction stabilises the C2S2 phase and leads to a subtle competition between all the different phases listed above. Indeed it is interesting to compare their Figure 8
with the full two-dimensional lattice model.
Wednesday, January 5, 2011
Curious people before bureacracy and buildings
Ahmed Zewail has a great Column in Nature, emphasizing the importance of basic science, and curiosity driven research for nation building. Here is an extract:
Curiosity-driven research requires that creative scientists work in an environment that encourages interactions between researchers and collaborations across different fields. But such attributes cannot and should not be orchestrated by structured and weighty management, as creative minds and bureaucracies do not work harmoniously together. So is there a formula for managing discovery making? The answer lies in accepting a triad of essentials.
First, and most important, are the people involved. Giving proper priority to providing thorough and inspiring education in science, technology, mathematics and engineering is essential. Research and development needs to attract the best young minds. Large buildings and massive funds will not produce much without the right people.
Second, an atmosphere of intellectual exchange is of paramount importance for ideas to crystallize. To distract faculty members with the writing of extensive and numerous proposals or to turn them into managers is the beginning of the end. The modern enterprise of science has become so bloated and complex that the traditional models of funding must be re-examined....
Third, without resources little can be achieved, no matter how creative the mind. Obviously, investment in science is needed to build instruments and to hire competent staff. Countries and institutions that provide the requisite infrastructure and the funding for ideas will be the homes of discoveries. But such support should follow the vision of creative researchers, not be built merely to lure money or to force people into fashionable research areas such as nanotechnology.
Monday, January 3, 2011
Molecular movies: watching excited state dynamics
Tomorrow I am visiting Ahmed Zewail's group at Caltech. This motivated re-reading a beautiful Science paper from 2005, Dark Structures in Molecular Radiationless Transitions Determined by Ultrafast Diffraction It contains the Figure below (which I have used on this blog and in talks before) of schematic potential energy surfaces for a ground and excited state.
A key issue in photochemistry and photophysics is how for a particular molecule one actually deduce such potential energy surfaces. Calculating them accurately from quantum chemistry is extremely difficult, even for small molecules. Trying to infer the topography of the surfaces from experiment is often ambiguous. Over the past decade Zewail's group has pioneered a powerful method for doing this. They optically excite the molecule and then monitor the molecular structure using ultra-fast electron diffraction.
A key issue in photochemistry and photophysics is how for a particular molecule one actually deduce such potential energy surfaces. Calculating them accurately from quantum chemistry is extremely difficult, even for small molecules. Trying to infer the topography of the surfaces from experiment is often ambiguous. Over the past decade Zewail's group has pioneered a powerful method for doing this. They optically excite the molecule and then monitor the molecular structure using ultra-fast electron diffraction.
Two key questions are addressed about the different photophysics of pyridine,
2-methylpyridine, and 2,6-dimethylpyridine.
What is the origin of the abrupt increase in the radiationless decay above a certain internal energy threshold [channel three threshold]?
How are the radiationless processes influenced by subtle changes in the molecular structure associated with the methyl groups?
The Figure below shows the structure of the three molecules after 50? psec. Note that the top two molecules [pyridine and methyl-pyridine] undergo ring opening whereas the third one [bi-methyl-pyridine] does not.
This observation is in stark contrast to the prevailing view that an ultrafast internal conversion pathway, mediated by the proximity of the first and second excited-state surfaces, opens up at the channel-three threshold and leads to vibrationally hot ground-state molecules.
It would be interesting to apply this technique to organometallic complexes that are used in LEDs. In particular, it should be possible to see the M-C bond rupture that has been proposed to be an important non-radiative decay channel.
Bottom up approach to rural electrification
There is a fascinating and inspiring story in the New York Times about how low cost solar cells and LED lighting is beginning to have an impact on some of the 1.5 Billion people in the world who live without electricity.
One of the companies involved Barefoot Power advocates a commendable bottom up approach to meeting basic needs in the developing world. [I was proud to see that the company has Australian origins].
One of the companies involved Barefoot Power advocates a commendable bottom up approach to meeting basic needs in the developing world. [I was proud to see that the company has Australian origins].
Sunday, January 2, 2011
Glueing together a theory
In elemental superconductors Cooper pairs form due to an attractive retarded electron-electron interaction that is mediated by exchange of phonons. A perennial and controversial question concerning superconductivity in strongly correlated electron materials (particularly the cuprates and organic charge transfer salts) is whether there is a similar "pairing glue". In a 2007 Science paper Phil Anderson claimed there is not.
A nice PRL by Maier, Poilblanc, and Scalapino addresses the question in a precise manner. The abstract has a nice summary of the issue.
The question of whether one should speak of a “pairing glue” in the Hubbard and t-J models is basically a question about the dynamics of the pairing interaction. If the dynamics of the pairing interaction arises from virtual states, whose energies correspond to the Mott gap, and give rise to the exchange coupling J, the interaction is instantaneous on the relative time scales of interest. In this case, while one might speak of an “instantaneous glue”, this interaction differs from the traditional picture of a retarded pairing interaction. However, as we will show, the dominant contribution to the pairing interaction for both of these models arises from energies reflecting the spectrum seen in the dynamic spin susceptibility. In this case, the basic interaction is retarded, and one speaks of a spin-fluctuation glue which mediates the d-wave pairing.
One thing that I found interesting and impressive about the paper is that it shows how one can define a dynamical pairing function in a non-perturbative manner. The relevant three equations are below. The Gorkov pairing (anomalous) Greens function F is defined by
which then leads to the pairing function
where G is the normal Greens function. This function has a real and imaginary part which can be related by Kramers-Kronig (or Cauchy) relations. The static part gives the pairing which can be written
The question regarding whether there is a “pairing glue” is then a question of whether the dominant contribution to φ1(kA,ω = 0) comes from the integral of φ2(kA, ω)/ω. From the results presented here we conclude that both the t-J and Hubbard models have spin-fluctuation “pairing glue”. However, they also exhibit a smaller, non-retarded contribution. For the cuprate materials, the relative weight of the retarded and non-retarded contributions to the pairing interaction remains an open question. Thus the continuing experimental search for a pairing glue in the cuprates is important and will play an essential role in determining the origin of the high Tc pairing interaction.
Deconstructing organometallic LEDs
A while back I posted Key questions about organometallic LED materials. Two of the questions were:
2. What is the non-radiative path from the emitting state? [i.e. what is the relevant vibrational co-ordinate?]
5. Is there a metal-centred (MC) excited state that is relevant to non-radiative decay?
[See also a Feature Article, Anthony Jacko, Ben Powell, and I wrote recently].
I just found a nice JACS paper, which comprises a combined theoretical and experimental study to help answer these questions. The abstract states:
2. What is the non-radiative path from the emitting state? [i.e. what is the relevant vibrational co-ordinate?]
5. Is there a metal-centred (MC) excited state that is relevant to non-radiative decay?
[See also a Feature Article, Anthony Jacko, Ben Powell, and I wrote recently].
I just found a nice JACS paper, which comprises a combined theoretical and experimental study to help answer these questions. The abstract states:
The kinetic parameters for decay and results from density functional theory (DFT) calculations of the triplet state are consistent with a nonradiative process involving Ir−N (Ir−C for fac-Ir(pmb)3) bond rupture leading to a five-coordinate species that has triplet metal-centered (3MC) character. Linear correlations are observed between the activation energy and the energy difference calculated for the emissive and 3MC states.The paper has several helpful figures such as the one below.
Tuning electronic dimensionality with chemistry
An interesting Chemistry of Materials paper illustrates how different types of stacking of organic molecules leads to electronic structures with different dimensionality. This is important in the quest to find Mott insulators with frustrated interactions that are tuned to produce a spin liquid ground state. However, it looks like these materials cannot be described by a half-filled band (there is only a small dimerisation) but rather should be described by a three-quarters filled band.
Saturday, January 1, 2011
New Annual Reviews journal in CMP
My subjective impression is that chemists are better at producing useful review articles, both in quality and quantity, than physicists. I particularly like Accounts in Chemical Research, Advances in Chemical Physics, Chemical Reviews, and Annual Review in Physical Chemistry. Hence, I was delighted to see that last year Annual Reviews started a new line, Annual Reviews in Condensed Matter Physics.
The Table of Contents of the first volume looks very promising.
The Table of Contents of the first volume looks very promising.
Subscribe to:
Posts (Atom)
Are chemical isomers emergent?
In discussions of emergence, particularly in chemistry, isomers are often given as an example of an emergent phenomenon. In Anderson's o...

-
This week Nobel Prizes will be announced. I have not done predictions since 2020 . This is a fun exercise. It is also good to reflect on w...
-
Is it something to do with breakdown of the Born-Oppenheimer approximation? In molecular spectroscopy you occasionally hear this term thro...
-
Nitrogen fluoride (NF) seems like a very simple molecule and you would think it would very well understood, particularly as it is small enou...