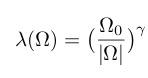There is no doubt that the world is changing very rapidly. This is true in many spheres: technology, politics, economics, and social. These changes present significant challenges to individuals, families, communities, businesses, institutions, and countries. On this blog there have been many posts and comments about how science and universities are changing.
I think there are three common mistakes in how people respond to these changes.
1. Denial. Claim that the changes are not really that significant (either qualitatively or quantitatively) and we should just keep on operating in the same way. This response will mostly come from those who are not directly affected in the short term.
2. On the other hand, some claim everything has changed and that everything is up for grabs, and they begin to lose sight of basic truths and goals, whether it is human aspirations or the content of physics curricula.
3. Seduction by the "change merchants." These are the opportunists: who want to use the change as a pretext to sell and implement their "solutions" from which they will increase their power, social status, or bank account.
Monday, September 24, 2018
Friday, September 14, 2018
Publishing for Majority World academics
Tomorrow I am giving a talk about academic publishing for a group of faculty and Ph.D students from African universities. The challenges they face are formidable.
Here are my slides.
As always, it is important not to reinvent the wheel.
There are already some excellent resources and organisations.
A particularly relevant organisation is AuthorAID which is related to inasp, and has an online course starting right now.
Publishing Scientific Papers in the Developing World is a helpful book, stemming from a 2010 conference.
Erik Thulstrup has a nice chapter "How should a Young Researcher Write and Publish a Good Research Paper?"
Here are my slides.
As always, it is important not to reinvent the wheel.
There are already some excellent resources and organisations.
A particularly relevant organisation is AuthorAID which is related to inasp, and has an online course starting right now.
Publishing Scientific Papers in the Developing World is a helpful book, stemming from a 2010 conference.
Erik Thulstrup has a nice chapter "How should a Young Researcher Write and Publish a Good Research Paper?"
Wednesday, September 12, 2018
Humility in science
Scientists often like to talk about how much they know and understand. On one level this is fine and appropriate because it is truly amazing how much we do now know and understand about the material world. Yet there are many things we don't really understand, and in some cases it may be argued we may never (at least in our lifetimes) understand certain things.
Furthermore, the preponderance of hype in science today tends to obscure and confuse what we don't understand. Humility can be a good thing for at least two reasons. First, it makes us more open to seeing our mistakes and misunderstandings. Maybe some things we think we do know and understand we may actually be wrong about. Second, in contrast to hype, humility helps us more clearly see and acknowledge the limitations of our current knowledge, so that we can explore ways forward.
Economics is an interesting case. My son, pointed out this quote from Hayek, a Nobel Laureate.
A concrete illustration of this ignorance is illustrating by considering the basic (and very important) question, "Does government stimulus spending actually produce economic growth?"
Economists cannot agree on the answer. This issue is nicely discussed in a podcast at Econtalk. Jim Manzi "argues for humility and lowered expectations when it comes to understanding causal effects in social settings related to public policy.''
The physical sciences are blessed with "controlled experiments" and the fact that physical systems seem to be a lot "simpler" than social systems. Nevertheless, that does not justify hype rather than humility.
Furthermore, the preponderance of hype in science today tends to obscure and confuse what we don't understand. Humility can be a good thing for at least two reasons. First, it makes us more open to seeing our mistakes and misunderstandings. Maybe some things we think we do know and understand we may actually be wrong about. Second, in contrast to hype, humility helps us more clearly see and acknowledge the limitations of our current knowledge, so that we can explore ways forward.
Economics is an interesting case. My son, pointed out this quote from Hayek, a Nobel Laureate.
“The curious task of economics is to demonstrate to men how little they really know about what they imagine they can design.”Friedrich A. Hayek, The Fatal Conceit: The Errors of Socialism
A concrete illustration of this ignorance is illustrating by considering the basic (and very important) question, "Does government stimulus spending actually produce economic growth?"
Economists cannot agree on the answer. This issue is nicely discussed in a podcast at Econtalk. Jim Manzi "argues for humility and lowered expectations when it comes to understanding causal effects in social settings related to public policy.''
The physical sciences are blessed with "controlled experiments" and the fact that physical systems seem to be a lot "simpler" than social systems. Nevertheless, that does not justify hype rather than humility.
Monday, September 10, 2018
What can students learn from an Ising model simulation?
Computer simulations can provide significant insight into different physical phenomena. Two decades ago the best one could do in a class or seminar was show screen shots of simulations and try and explain what was going on. Now one can show a simulation live and even vary parameters in real time to provide insight. I have done this quite a bit with Solid State Simulations.
One simulation I like but have never used effectively is that of the Ising model.
See for example, Daniel Schroeder's simulation or James Sethna or Matt Bierbaum.
What does it help me understand?
The main ideas are the concept of symmetry breaking, the correlation length, and the divergence of the correlation length at the critical point.
1. Watching the different configurations changing with time illustrates the notion of an ensemble.
2. At high temperatures one sees the paramagnetic phase where the spins are independent of each other and so there are no domains.
3. As the temperature approaches the critical temperature (T=2.27J) from above the correlation length increases and large fluctuating domains form.
4. Below the critical temperature large domains form and fluctuate less and less as the temperature lowers.
5. The ferromagnetic ground state (blue or yellow, up or down spin) in zero external field depends on the history. This illustrates symmetry breaking
Any other things?
One simulation I like but have never used effectively is that of the Ising model.
See for example, Daniel Schroeder's simulation or James Sethna or Matt Bierbaum.
What does it help me understand?
The main ideas are the concept of symmetry breaking, the correlation length, and the divergence of the correlation length at the critical point.
1. Watching the different configurations changing with time illustrates the notion of an ensemble.
2. At high temperatures one sees the paramagnetic phase where the spins are independent of each other and so there are no domains.
3. As the temperature approaches the critical temperature (T=2.27J) from above the correlation length increases and large fluctuating domains form.
4. Below the critical temperature large domains form and fluctuate less and less as the temperature lowers.
5. The ferromagnetic ground state (blue or yellow, up or down spin) in zero external field depends on the history. This illustrates symmetry breaking
Any other things?
Wednesday, September 5, 2018
Superconductivity in a Hund's metal
The BCS theory of superconductivity is one of the towering intellectual achievements of the twentieth century. There are many ingredients to the theory and many significant results. One key step is to consider an effective interaction that is responsible for the Cooper pairing. A key result is that many properties are universal in that one can rescale temperatures and energies by the energy gap (at zero temperature), Delta(0) or the transition temperature Tc. In the limit of weak-coupling there is a universal ratio
2 Delta(0)/kTc = 3.5
Most elemental superconductors are consistent with this value. Some such as Hg and Pb have larger values, but these can actually be calculated when strong coupling effects are taken into account, via the Eliashberg equations.
Unconventional superconductors (cuprate, organic, heavy fermion, iron based) have resisted a simple unifying theory and universal trends, comparable to the stellar success of BCS theory. For example, the gap/Tc ratio is all over the place. However, there has been some progress for the iron-based superconductors. Recent ARPES results (summarised in the figure at the bottom below) have shown a universal ratio, of about 7.2 for a wide range of materials.
A fascinating feature of these iron-based materials is the nature of the metallic state that undergoes the superconducting instability. I have written several blog posts about the Hund's metal. One important feature is that there is relatively low coherence temperature below which a Fermi liquid metal forms, and there is a correspondingly low energy scale Omega0 associated with spin fluctuations, which become very slow. This arises from the rich Kondo physics associated with the multi-orbital character of the system. Furthermore, the spin fluctuation spectrum has a power law dependence above Omega0.
The above ideas come together in an interesting preprint
On the Superconductivity of Hund's Metals
Tsung-Han Lee, Andrey Chubukov, Hu Miao, Gabriel Kotliar
One thing should be stressed here is that one is observing a transition from an incoherent metal into a superconductor, unlike in the BCS situation where the transition is from a coherent Fermi liquid.
I thank Alejandro Mezio for bringing the paper to my attention.
2 Delta(0)/kTc = 3.5
Most elemental superconductors are consistent with this value. Some such as Hg and Pb have larger values, but these can actually be calculated when strong coupling effects are taken into account, via the Eliashberg equations.
Unconventional superconductors (cuprate, organic, heavy fermion, iron based) have resisted a simple unifying theory and universal trends, comparable to the stellar success of BCS theory. For example, the gap/Tc ratio is all over the place. However, there has been some progress for the iron-based superconductors. Recent ARPES results (summarised in the figure at the bottom below) have shown a universal ratio, of about 7.2 for a wide range of materials.
A fascinating feature of these iron-based materials is the nature of the metallic state that undergoes the superconducting instability. I have written several blog posts about the Hund's metal. One important feature is that there is relatively low coherence temperature below which a Fermi liquid metal forms, and there is a correspondingly low energy scale Omega0 associated with spin fluctuations, which become very slow. This arises from the rich Kondo physics associated with the multi-orbital character of the system. Furthermore, the spin fluctuation spectrum has a power law dependence above Omega0.
The above ideas come together in an interesting preprint
On the Superconductivity of Hund's Metals
Tsung-Han Lee, Andrey Chubukov, Hu Miao, Gabriel Kotliar
They consider a single band superconductor described by the strong-coupling Eliashberg equations where the frequency dependence of the (effective) electron-electron attraction is given by
where the exponent gamma is treated as a variable. The Eliashberg equations are solved (for a single band) and give the following relationship between the gap ratio and the exponent gamma.
The value of gamma=1.2 is that associated with the relevant Kondo problem above the coherence temperature. The gap ratio corresponds to the black dashed line in the graph below.One thing should be stressed here is that one is observing a transition from an incoherent metal into a superconductor, unlike in the BCS situation where the transition is from a coherent Fermi liquid.
I thank Alejandro Mezio for bringing the paper to my attention.
Monday, September 3, 2018
Absurd to the n'th degree, not chemical cricket
It is easy to take for granted many scientific ideas and results that seem commonplace and reasonable to us today. However, when some ideas were first proposed or discoveries made they were greeted with scepticism, and even ridicule.
In 1913 the Braggs determined the crystal structure of sodium chloride. They were awarded the Nobel Prize in Physics in 1915. However, as late as 1927, the following letter appeared in Nature.
[Double click if you want to read a larger version].
I recently became aware of this in Crystallography: A Very Short Introduction by A.M. Glazer.
In 1913 the Braggs determined the crystal structure of sodium chloride. They were awarded the Nobel Prize in Physics in 1915. However, as late as 1927, the following letter appeared in Nature.
[Double click if you want to read a larger version].
I recently became aware of this in Crystallography: A Very Short Introduction by A.M. Glazer.
Subscribe to:
Posts (Atom)
The emergence of molecular structure from quantum theory
Most debates about the emergence of molecular structure centre around the issue of irreducibility. Specifically, can the existence of molecu...

-
This week Nobel Prizes will be announced. I have not done predictions since 2020 . This is a fun exercise. It is also good to reflect on w...
-
Is it something to do with breakdown of the Born-Oppenheimer approximation? In molecular spectroscopy you occasionally hear this term thro...
-
Nitrogen fluoride (NF) seems like a very simple molecule and you would think it would very well understood, particularly as it is small enou...