So, what is the main thesis of your Ph.D? Are you ready to defend this thesis?
Tuesday, June 29, 2010
A thesis should have a thesis
Yes. I heard a humanities scholar speak about this today. Indeed, according to Wikipedia, "the word "thesis" comes from the Greek θέσις, meaning "position", and refers to an intellectual proposition."
Monday, June 28, 2010
The challenge of energy sustainability
Today at the I-CAMP School in Brisbane, Dan Cox, Director of I2CAM gave a nice talk about climate change and future energy options.
Dan has taught an undergraduate course on such issues for the past 13 years at UC Davis. (Unfortunately, the course web page seems to be down right now).
He mentioned some really nice lectures on the same topic by Nate Lewis (Caltech)
A few things I found interesting.
Graphs of energy consumption vs. GDP tend to plateaus at high incomes. i.e, affluent countries can increase their "standard of living" without increasing their energy consumption. Japan and EU use about half the energy per capita of USA/Canada, yet have comparable standards of living.
Electricity consumption is 4 times greater in Wyoming than in California.
Oil is used almost 100% for transportation. Alternative electricity sources will not reduce dependence on Middle East oil without electric cars.
M. King Hubbert was a petroleum engineer who predicted world oil production would peak in 1970. The production rate is related to the derivative of the solution to the logistic differential equation, which describes many phenomena that initially increase exponentially until finite resources limit their growth.
[Aside there was actually a West Wing episode, The Hubbert Peak]
CO2 dissolving in ocean decreases pH and destroys coral. Even if global temperatures do not increase due to increased CO2, the increase in acidity of the oceans will wreak environmental havoc.
Searching for better solar cell dyes
This morning I looked over a nice review article on Metal free organic dyes for dye-sensitized solar cells. Here are a few highlights and comments.
The figure below is a schematic of what one is trying to design: a dye molecule D-pi-A in which upon excitation by a photon the excited state (D-pi-A)* decays non-radiatively into a charge separated state D+pi-D- which leads to charge injection into the TiO2.

Many of the dyes have the structural motif of methine dyes and are prone to cis-trans isomerisation in their excited state. It is implied that this is a bad thing one wants to stop. I actually thing an optimised/directed isomerisation could be good because it can lead to a twisted intramolecular charge transfer (TICT) state.
I am curious to what extent many of these dyes can be described by the type of effective Hamiltonians that Seth Olsen and I (and also independently by Anna Painelli and collaborators) have been exploring for other methine dyes. This could help answer questions about TICT states and design principles.
The following summary point by the authors is noteworthy:
It is therefore important to determine design rules for organic dyes to achieve improved properties and performance so that they may in the future compete or supersede ruthenium(II) sensitizers. Clearly, the development and optimization of materials for organic solar cells in general is not (yet) rational, but rather empirical. This is due to the multiple parameters which have to be taken into account when novel dyes and materials are designed for organic solar cells. The efficiency of the solar cells does not only depend on the molecular structure of the dye, but sometimes even more so on solid-state properties, such as aggregation, morphology, and self-assembly. In the case of DSSCs, the efficiency additionally depends on the type of photoelectrode, anchoring group of the dye, electrolyte, and mediating redox couple.
To me it raises questions about whether
-so much effort should be expended on making new dyes rather than on understanding solid state interactions.
-authors should be more cautious about attributing variations in device efficiency to properties of specific dyes rather than solid state properties.
Saturday, June 26, 2010
Theoretical chemistry in the Rockies
Apparently chemists got jealous of the Aspen Center for Physics and so started the Telluride Science Research Center.
I am looking forward to going there in three weeks for a workshop on Condensed Phase Dynamics. It will be great because I will get to meet some theoretical chemists whose papers I have read but not met yet, including Garnet Chan, Phillip Geiser, Dimitrii Makarov, Bill Miller, Peter Rossky, Greg Voth, .....
Friday, June 25, 2010
Quantifying the effect of "small" chemical changes
Physicists say the details don't matter. Chemists say they do. Biologists say the details are a matter of life and death.
If you make a "small" change in a molecule what effect will it have on its properties?
Long ago Hammett found a fairly robust and empirical way [the Hammett equation] to quantify the answer, at least for organic chemical reactions involving aromatic molecules.
A nice J. Phys. Chem. A paper by Cordes et al. considers the following important problem in photophysics. Consider the molecule below which upon irradiation can undergo a conformational change (photoisomerisation).
If the substituents R1 and R2 are changed what effect does that have on the rate of photoisomerisation (non-radiative decay)?

They find the rate of both the forward and backward photochemical reactions [which varies by two orders of magnitude] can be correlated with Hammett's parameters for the substituents.
Wednesday, June 23, 2010
Fermions or bosons?
In a "round table discussion" about the theory of the cuprate superconductors Patrick Lee suggested that the genuinely new idea that has been developed is:
"the notion of emergence of gauge fields and fractionalized particles as low-energy phenomena in systems that did not contain them in the starting model."
He suggests that this idea is of comparable importance in condensed matter theory to that of Goldstone bosons.
Gauge fields emerge when the electron or spin operators are represented in an alternative manner such as in terms of Schwinger bosons, slave fermions, slave bosons, or slave rotors. But a key question is for a given model Hamiltonian, which is the appropriate representation.
For quantum spin models it seems that which side of the Charles River you work on determines your preference for a particular representation. At Harvard, Subir Sachdev favours bosonic spinons, while on the opposite of the river, at MIT Patrick Lee favours fermionic spinons.
Where do the gauge fields come from?

Tuesday, June 22, 2010
Exam post mortem
This week I marked the exams for the undergraduate course I helped teach this past semester. I usually find this a depressing exercise. It painfully reveals how little I have managed to teach students and how little they have learnt. Note, I consider the responsibility is joint.
The last three questions in the exam are from the condensed matter part of the course I teach. The students have seen the questions before, either in tutorial problems, assignments, or past exam papers. Yet they struggle to do them.
Some of the questions are meant to test not just knowledge but also general skills such as converting units, summarising results in a figure, and sketching curves. But, this is what I find particularly disappointing. Students still have trouble doing some of these things, even at second year university.
I think next time I teach the course I particularly need to keep hammering home that all physical quantities have units, and that you need to keep track of them in calculations. Furthermore, I want the tutors to take off marks on assignments when students don't properly keep track of units.
Monday, June 21, 2010
How does protein structure optimise function?
It actually has something to do with strong electronic correlations and the Jahn-Teller effect. Furthermore, it illustrates chemistry is very local.
There is a nice review article, Molecular modelling for transition metal complexes: Dealing with d-electron effects by Deeth, Anastasi, Diedrich, and Randell. It discusses specific examples of the Jahn-Teller effect in d9 Cu(II) complexes. [Aside: this the same complex as in cuprate superconductors].
Type I blue copper proteins are involved in electron transfer in both plant and bacterial photo-synthesis. A key question concerns how the structure around the Cu active site might be optimised in order to facilitate electron transfer. It was proposed that the local environment was arranged to minimise the reorganisation energy in the Marcus-Hush electron transfer theory. However, using quantum chemistry calculations, Ryde, Olssen, Pierloot, and Roos, showed that this was not the case in 1996. In fact the local geometry was the same in vacuo as in the protein. Furthermore, the structure was dominated by the strong Cu-thiolate bond.

Hence, Deeth et al., state
We begin with the presumption that although protein molecules may be large and complex, they form simple M–L coordinate bonds – i.e. a Cu–N(imidazole) bond can be handled in exactly the same way irrespective of whether it corresponds to an isolated imidazole or to a histidine.
This allows them to develop force fields for molecular mechanics.
Chemistry is local. This is why localised approaches such as valence bond theory are often preferable to molecular orbital or DFT-based approaches which tend to delocalise electrons.
Classification of spin liquids
A paper in need to understand better is a 2002 PRB by Wen.
Wen used quantum orders and projective symmetry groups, to construct hundreds of symmetric spin liquids, having either SU(2), U(1), or Z2 gauge structures at low energies. He divided the spin liquids into four classes:
(a) Rigid spin liquid—spinons (and all other excitations) are fully gapped and may have bosonic, fermionic, or fractional statistics.
(b) Fermi spin liquid—spinons are gapless and are described by a Fermi liquid theory (i.e, the interaction between quasiparticles on the Fermi surface vanishes.)
(c) Algebraic spin liquid—spinons are gapless, but they are not described by free fermionic-bosonic quasiparticles.
(d) Bose spin liquid—low-lying gapless excitations are described by a free-boson theory.
Saturday, June 19, 2010
The Art of Being a Scientist
The latest Physics Today has a positive review by Renee Diehl of a new book, The Art of Being a Scientist: A Guide for Graduate Students and Their Mentors, by Roel Sineder and Ken Larner.
Friday, June 18, 2010
Effective Ph.D supervision
I mentioned previously that my colleague Ben Powell went to a helpful workshop on supervising Ph.D students. Stimulated by this he sent an email to all his students and postdocs with the following requirements for future meetings.
It should contain:
- Your report on what you have done in since our last meeting
- An agenda for the meeting. This MUST include the following items
(but may include more)
- discussion of what you have done
- discussion of what you will do next
- one tangible observable that everyone at the meeting agrees you are going to do next
- Any upcoming deadlines (even if they are far away)
- Something you have (formally) written since the last meeting (perhaps a summary of a paper you read, a draft of a thesis chapter or paper, details of your latest calculations)
2) At the meeting we should take time for each of items on the agenda.
3) Within 6 hours of the end of the meeting you should send me another email. It should contain:
- A summary of the meeting
- What you tangible observable for the next week is
- A to do list (this should be copied, pasted and edited from your previous after-meeting report so that things don't get dropped off the list). At least near the top of the list, it should be in priority order.
Somewhat related issues concerning student-supervisor expectations are discussed in an old post I wrote.
Thursday, June 17, 2010
An unexpected twist
Lately I have become somewhat obsessed with trying to find concrete examples of conical intersections between potential energy surfaces of excited states of organometallic complexes. One example is here.
This led to reading a nice paper in Journal of Physical Chemistry Letters by Zurek and Paterson. They consider a platinum complex which undergoes a significant structural change upon photoexcitation (photoisomerisation).
However, contrary to what was thought originally the metal is just a spectator. In particular, the relevant excited states are not MLCT (metal-to-ligand charge transfer) states but rather pi-pi* states localised on the organic ligand.

Radiationless deactivation does occur through a conical intersection, but this intersection is also present in just the ligand. The key motion is associated with the C-N-C bond which links the two quinolone parts of the ligand. The intersection occurs when the CNC angle is 120 degrees and the two quinoline groups are twisted almost 90 degrees.
One question I have is what is the simplest possible effective Hamiltonian which could capture this photophysics.
Wednesday, June 16, 2010
The goal of physics undergraduate courses
I do not think the goal should be train or recruit Ph.D students to work on our favourite projects. Most students who take undergraduate courses will not end up doing Ph.Ds or in research careers. These students should be our focus. I hope they will learn a range of concepts, approaches, and skills that will equip them for a range of future opportunities in life.
Key skills students can (and should) learn in physics courses are:
- to solve problems
- to make orders of magnitude estimates
- to think and work quantitatively
- to think critically and evaluate truth claims
- to design and perform experiments and analyse data
- to perform error analysis
- to perform dimensional analysis
- to make approximations and estimate their validity
The article Teaching Biological Physics discusses some of these issues.
Perhaps, undergraduate physics courses should not be viewed as a "professional degree" but rather from the perspective of a liberal arts education. A book I really like and mentioned previously is Five Minds for the Future, by Howard Gardner.
Quantum effects and noise in biomolecules
New Journal of Physics has a Focus issue on Quantum Effects and Noise in Biomolecules. This includes a paper by myself and my former students Jacques Bothma and Joel Gilmore, The role of quantum effects in proton transfer reactions in enzymes: quantum tunneling in a noisy environment?
Tuesday, June 15, 2010
Fluorescent tonic water
Monday, June 14, 2010
Justifying Effective Hamiltonians
Physicists like effective Hamiltonians such as the Hubbard, Heisenberg, and Anderson models. Why do they work so well? What do the parameters in the Hamiltonian actually correspond to? How are the orbitals related to atomic and molecular orbitals?
These days, computational chemists are not so enthusiastic about such semi-empirical theories.
Today I read through a nice article, Is There a Bridge between ab Initio and Semiempirical Theories of Valence?, by Karl Freed.
It describes a strategy for partitioning the full Hilbert space into two parts P and Q, where P is the space of configurations involving only valence electrons.
The key equation (8) gives an exact expression for the valence Hamiltonian [which acts only on the space of states P] in terms of the full Hamiltonian.
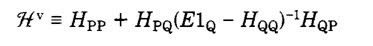
He then explicitly evaluates the Hamiltonian matrix elements for small atoms and molecules. The values are consistent with experiment but significantly different from those evaluated at the Hartree-Fock (self-consistent field) level.
Saturday, June 12, 2010
A successful Ph.D
This week at UQ there was a workshop on supervising Ph.D students that my colleague, Ben Powell went to. He said it was very helpful. It was run by ThinkWell. It looks like they have some really good material. Later I will write more about something Ben learnt about running more effective student-supervisor meetings.
Here is the Table of Contents of their book.

- Care and maintenance of your supervisor
- Write and show as you go: This is show and tell not hide and seek
- Be realistic: It's not a Nobel Prize
- Say no to distractions: Even the fun ones and the ones you think you must do
- It's a job: That means working nine to five but you get holidays
- Get help: You are not an owner-operator single person business
- You can do it: A PhD is 90% persistence and 10% intelligence
Certainly looks like worth reading.
Thursday, June 10, 2010
Basics of organic photonics
Today I am giving the "cake meeting" talk. I will mostly go through some key ideas about excited states of organic molecules, that I mentioned in the previous post.
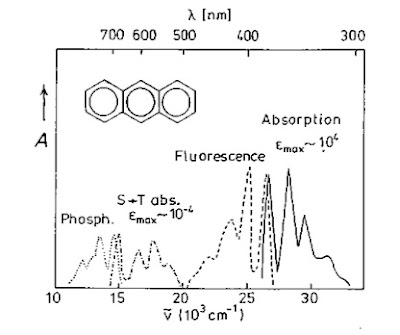
The figure below nicely illustrates a few key ideas.
It shows the absorption and emission spectrum of anthracene.
The high energy feature is associated with the lowest "bright" singlet state.
The low energy feature is associated with the lowest triplet state.
Note
- the vibrational satellites (these are mostly aromatic C-C stretches)
- the "mirror-image" symmetry of the absorption and emission spectrum.
- the singlet absorption is 8 orders of magnitude larger than the triplet
- the large singlet-triplet energy gap
I will then flag how the full machinery of quantum many-body theory can attack some (but far from all) of these issues: vibronic structure, mirror-image rule, Huang-Rhys factor, Stokes shifts....
The relevant formalism is contained in a section of Mahan's tome, Many-Particle Physics. It allows one to treat many-modes, temperature dependence, and strong coupling, ...
Wednesday, June 9, 2010
Re-inventing the wheel (with open boundary conditions...)
The Journal of Chemical Theory and Computation just published a paper,
Conjugated Molecules Described by a One-Dimensional Dirac Equation
I had a sense of deja vu reading the first two-thirds of the paper. In the 1980's there was a lot of work in this direction in the physics community, beginning with this PRB by Takayama, Liu, and Maki, which has hundreds of citations. However, the JCTC paper authors appear to be unaware of all this earlier work.
Writing credible grant applications
Is your proposal credible?
For example, even IF you develop the correct theory of cuprate superconductivity it is not a given it is going to lead to levitating trains and a new electrical power distribution network.
The past is a pretty good predictor of the future. The quality and quantity of your output, and the impact of your research, is not likely to change that dramatically, even if you get the funding.
For example, if in the past 5 years you have not done anything particularly significant then it is hard for me to believe you will if you get this grant. On the other hand, if your grants always lead to solid publications that generate interest, then it is a pretty safe bet that another grant will.
Furthermore, if there is some problem that many other smart, technically able, and well funded people in the world are struggling to solve an important problem, it is hard to believe that YOU are going to solve it.
Tuesday, June 8, 2010
Deconfined spinons take the heat
There is a nice paper in Science this week which provides experimental evidence for gapless spin excitations (possibly deconfined spinons) in an organic material that may have a (Mott insulating) spin liquid ground state.
[I thank Andrew Bardin and Ben Powell for bringing it to my attention].
The Figure below shows the temperature dependence of the thermal conductivity kappa(T). The fact that as T->0, kappa/T has a non-zero intercept is a clear signature of gapless excitations. The magnitude of the intercept is comparable to its value in the metallic phase of other organic charge transfer salts, and an order of magnitude larger than what one gets in the d-wave superconducting state due to nodal quasi-particles. [see for example this PRL].

[This is a good example of a Figure because it compares results for several materials so you can see just how clearly different the dmit-131 material is].
The authors also measure the thermal conductance tensor in a magnetic field. Within error the thermal Hall angle was zero. This was motivated by this recent PRL by Katsura, Nagaosa, and Lee, predicted a sizeable thermal Hall effect in quantum spin liquids with deconfined spinons. [Patrick Lee wrote a guest post about this a while back].
Monday, June 7, 2010
What value is this paper?

I finally read Conyer Herring's article Distill or Drown: the need for reviews.
It contains the figure above which is worth thinking about.
It provides a nice classification of the value of published papers.
Furthermore, it claims the distribution in value shifts with time, so that after 5 years most papers are not of significant value.
Saturday, June 5, 2010
20 key concepts in thermodynamics & condensed matter
Today I gave my last lecture for the second year undergraduate course in thermodynamics and condensed matter. I gave a summary of the whole course!
In the first part I try to show the best logical structure of thermodynamics is to actually start with the Second law. This was something I was introduced to as an undergraduate by Hans Buchdahl (who later became my honours supervisor). His ideas are contained in his The Concepts of Classical Thermodynamics. Similar ideas are now championed by Elliot Lieb and Jacob Yngvason, and described in this nice Physics Today article.
Friday, June 4, 2010
The character of the spin liquid ground state in some real materials
This morning I read an interesting preprint, Weak Mott insulators on the triangular lattice: possibility of a gapless nematic quantum spin liquid, byTarun Grover, N. Trivedi, T. Senthil, Patrick A. Lee
They study the ground state of Heisenberg model on the isotropic triangular lattice with ring exchange. This model is relevant to the Mott insulating phase of two different organic charge transfer salts which may have a spin liquid ground state.
The authors perform variational Monte Carlo for Gutzwiller-projected BCS states. (These are RVB=resonating valence bond states, proposed by P.W. Anderson in 1987). A mean-field theory on these states gives a BCS state with broken time-reversal symmetry (known as the chiral spin liquid). The form of the fermion pairing function is d+id (d_x^2-y^2 + i d_xy) which belongs to the E representation of the C_6v point group symmetry of the lattice.
A few highlights from the paper:
- For a range of ring exchange strengthes (0.1
- The authors suggest that under pressure (increasing t/U) the Mott insulator will be destroyed leading to a superconducting state with the same d_x^2-y^2 pairing. This is qualitatively different to what one gets with a mean-field RVB theory of the model without the ring exchange (described in this PRL by Ben Powell and myself).
- The theory cannot explain why the observed low temperature specific heat of kappa-ET2-(CN)3 is weakly dependent on magnetic field.
- The "Amperean pairing" theory proposed earlier by Lee, Lee, and Senthil does not have this problem but has difficulty describing the superconducting state which develops under pressure.
Several issues not addressed in the paper are:
1. Exact diagonalisation calculations on small lattices (Ref. 10) give a different spin liquid ground state, one with an energy gap to triplet excitations, and many singlet excitations inside the gap.
2. The NMR relaxation rate 1/T_1 for kappa-ET2-(CN)3 has a power law temperature dependence consistent with gapless excitations.
3. The estimate of the impurity scattering rate (~1.5 K) can be compared to independent estimates see other organic charge transfer salts. (See Table I in this PRB, I think the estimates there suggest the authors proposal that the scattering rate may be even larger than their estimate is unlikely).
4. The real materials may have a small spatial isotropy. The Heisenberg model on the anisotropic triangular lattice can also have a spin liquid ground state, without the need for ring exchange (see for example, this paper).
Wednesday, June 2, 2010
Hans Buchdahl (1919-2010): his legacy
Hans Buchdahl got me started in theoretical physics. When I was an undergraduate he was the Professor of Theoretical Physics at the Australian National University. He was the supervisor of my honours year project, which led to my first two papers. Buchdahl taught me many things including:
- theoretical physics is fun, exciting, and hard work
- the beauty of classical thermodynamics
- the importance of conceptual rigour
- high standards
- that I had something to contribute
This week I learnt of his death, earlier this year. There is a nice Obituary in the Canberra Times and Sydney Morning Herald by John Sandeman and one in General Relativity and Gravitation by Hubert Goenner.
To complement those I should point out a significant legacy that Buchdahl left in terms of the undergraduate students that he inspired and shepherded and have gone onto successful academic careers. Besides myself, a few that I know of who did an honours year project supervised by Buchdahl include:
Professor of Theoretical Physics,
Head of Department of Physics
Queen Mary College London
Simons Professor of Mathematics
MIT
Professor of Physics and Mathematics
University of Southern California
Buchdahl was always concerned with what would be best for students and not himself. He strongly encouraged us to go to the best places in the world for our Ph.Ds. I am sure that the letters of reference that he wrote on our behalf helped make that possible.
One more random fact. When Buchdahl retired in 1984 the ANU did not consider that his Chair should be replaced. No doubt to save money they absorbed the Theoretical Physics department into Physics and advertised for a replacement for Buchdahl at the Lecturer level. This position was filled by a young Gerard Milburn. He left after a few years to come to University of Queensland, where he has built a great place to work!
Tuesday, June 1, 2010
Want to improve student learning? Then fail more!
Six months ago I mentioned an excellent post by the Female Science Professor about the struggle of teaching graduate students to write. There are a lot of perceptive and helpful comments on that post. There are two comments which have stayed with me and I wish to highlight because they are relevant to much broader issues in undergraduate education.
We all need to improve the quality of our teaching. But sometimes I think the best thing we could do for our students is fail more of them. Furthermore, as one of the comments above notes dealing with failure (or at least the threat of it) is important preparation for life in the "real world."
Linda said... I don't believe that this is a skills issue.Currently I am teaching a second year undergraduate course on thermodynamics and condensed matter. To enrol students should have passed a course on multi-variable calculus and a first year calculus based physics course. Yet there are many students in the class who struggle to do basic things like:
The issue is that there are no consequences for writing badly. That student, one way or another (including buying papers), will continue on in his/her education.
When we take this seriously, students will be told "no, you cannot continue on in your education until you improve this skill".
Oh, the horror! Holding students (at ANY level) to a standard. Isn't that unconstitutional or something?
Anonymous said...
My English 101 writing teacher threatened to fail me unless I improved. My paper was,
- too long ... 5x requested length
- contained too many parenthesis
- contained too many footnotes
- thesis unclear
- etc.
I made those improvements. Compared to professional writers I know I could improve much more. Still, the professor made her point.
What's difficult to understand here? Fail the student if the writing under performs.
I attended university in my mid-30s. I applied the basic principles operating in most work environments. Those in charge identify areas of weakness and require improvement in their subordinates. And either the worker improves or risks being replaced. Ideally this improvement is done with the cooperation and support of the boss.
- Sketch a graph of a one variable function.
- Find the value of x at which a specific function is a minimum.
- Evaluate a partial derivative of a simple function of more than one variable.
- Keep track of physical units in a calculation.
We all need to improve the quality of our teaching. But sometimes I think the best thing we could do for our students is fail more of them. Furthermore, as one of the comments above notes dealing with failure (or at least the threat of it) is important preparation for life in the "real world."
Subscribe to:
Posts (Atom)
What is condensed matter physics?
Every day we encounter a diversity of materials: liquids, glass, ceramics, metals, crystals, magnets, plastics, semiconductors, foams, … Th...
-
This week Nobel Prizes will be announced. I have not done predictions since 2020 . This is a fun exercise. It is also good to reflect on w...
-
Is it something to do with breakdown of the Born-Oppenheimer approximation? In molecular spectroscopy you occasionally hear this term thro...
-
Nitrogen fluoride (NF) seems like a very simple molecule and you would think it would very well understood, particularly as it is small enou...



