I previously posted about how double proton transfer is a concrete example of a chemical reaction that can occur via either a concerted or sequential process?
Precisely defining this question and answering it is a subtle issue.
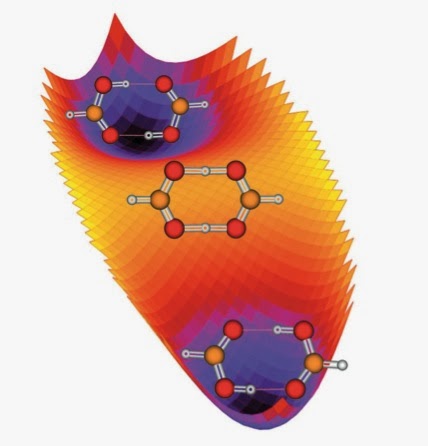
There is a nice classification of types of potential energy surfaces for double proton transfer, summarised on the website of Antonio Fernandez-Ramos. It is based on a very simple model potential energy surface described here and compared to surfaces from computational quantum chemistry [at the DFT level] here [source of the figures below].
There are three qualitatively different potential energy surfaces, depending on the strength of the coupling of the motion of the two protons.
(1) One transition state and two minima, as in the formic acid dimer;
(2) Two equivalent transition states, one maxima and two minima, as in the pyrazole dimer;
(3) Four transition states, one maxima and four minima, as in porphine.
I am pretty happy because I am developing a simple diabatic state model that captures all of the above cases.
Subscribe to:
Post Comments (Atom)
What Americans might want to know about getting a job in an Australian university
Universities and scientific research in the USA are facing a dire future. Understandably, some scientists are considering leaving the USA. I...
-
Is it something to do with breakdown of the Born-Oppenheimer approximation? In molecular spectroscopy you occasionally hear this term thro...
-
Nitrogen fluoride (NF) seems like a very simple molecule and you would think it would very well understood, particularly as it is small enou...
-
I welcome discussion on this point. I don't think it is as sensitive or as important a topic as the author order on papers. With rega...




No comments:
Post a Comment