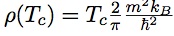H-bond correlations and NMR chemical shifts
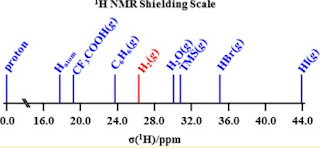
For a diverse range of chemical compounds, the strength of hydrogen bonds [parametrised by the binding energy and/or bond length] is correlated with a wide range of physical properties such as bond lengths, vibrational frequencies and intensities, and isotope effects. I have posted about many of these and a summary of the main ones is in this paper. One correlation which is particularly important for practical reasons is the correlation of bond strength (and length) with the chemical shift associated with proton NMR. The chemical shift is the difference between the NMR resonant frequency of the proton in a specific molecule and that of a free proton. The first important point is that although this shift is extremely small (typically one part in 100,000!) one can measure it extremely accurately. More importantly, this shift is quite sensitive to the local chemical bonding and so one can use it to actually identify the bonding in unknown molecules (e.g. protein structure determin